Dual Membrane Passive Diffusion Bag Study for Per- and Polyfluoroalkyl Substances

Originally published in The Military Engineer, Vol. 113, No. 734, July-August 2021
Due to the historical use of aqueous film forming foam (AFFF) with per- and polyfluoroalkyl substances (PFAS), the Department of Defense (DoD) is currently managing a growing number of sites with known or suspected PFAS contamination. Groundwater monitoring for PFAS at these installations is conducted using low-flow well sampling procedures, which employ portable pumps, dedicated tubing, real-time water quality measurements and may require management of investigation-derived waste (IDW).
A research team comprising EA, EON Products, Inc., and the Omaha District of the U.S. Army Corps of Engineers evaluated the effectiveness of dual membrane passive diffusion bag (DMPDB) technology to collect representative groundwater samples for PFAS analysis. Although passive sampling groundwater technologies have been routinely used for more than 20 years at DoD sites for volatile organic compounds, metals, and inorganic analysis, their reliability for monitoring PFAS in groundwater is subject of ongoing bench-scale and field study research.
Current Alternatives
There are several types of passive samplers currently under evaluation for the collection of groundwater samples for PFAS analysis. These include grab samplers for instantaneous recovery; sorption-type samplers where contaminant mass accumulated over a known duration is used to calculate concentration; and equilibrium-type samplers that reach and maintain equilibrium with the sampled medium.
Traditional PDBs consist of a semi-permeable membrane bags filled with deionized water. The bags are placed in the saturated screen intervals of groundwater monitoring wells where molecules diffuse across the membrane pores into the sampler until the concentration equalizes between the groundwater and the inside of the sampler. The single polyethylene diffusive membrane utilized in traditional PDBs is permeable to non-polar volatile organic compounds but not to other common analytes.
It has been well documented that the collection of groundwater samples using passive samplers results in significant cost and time savings relative to other common sampling techniques, due to lower equipment and labor costs. Passive samplers also lower the chance of cross-contamination, allow for depth-specific profiling, and generate less investigation derived waste. This is of particular concern at PFAS sites where IDW treatment and disposal techniques are still evolving. Additionally, several passive sampling technologies yield very low turbidity sample aliquots, which is vital for reduced matrix interference during laboratory analysis. Equilibrium-type samplers also allow for sample aliquots to be collected directly in the field and prepared for analysis using conventional methods. Conversely, sorptive samplers require additional preparatory work to extract target chemicals from the sorptive media prior to analysis.
Dual Membrane Passive Diffusion Bag Technology Description
DMPDBs apply the same concept as PDBs but utilize two membranes with different diffusion capabilities to expand the analyte list beyond volatile organic compounds. None of the DMPDB materials are considered to be sources of PFAS. The upper membrane, made of polyamide, has larger pores and is hydrophilic. This facilitates diffusion of polar and larger molecules into the sampler, such as metals, cations, anions, and 1,4-dioxane. PFAS, which consist of carbon chains bonded to fluorine atoms with hydrophilic polar functional groups, are both polar and relatively large compared to volatile organic compounds. They pass through the upper membrane of the DMPDBs. The lower membrane, made of high-density polyethylene, has smaller pores and is hydrophobic. It is permeable to relatively small volatile organic compound molecules.
This lower membrane also prevents water from escaping the sampler, serving as a sample reservoir. Suspension tethers are typically made of polypropylene-braided cord manufactured without PFAS and are dedicated to each well.
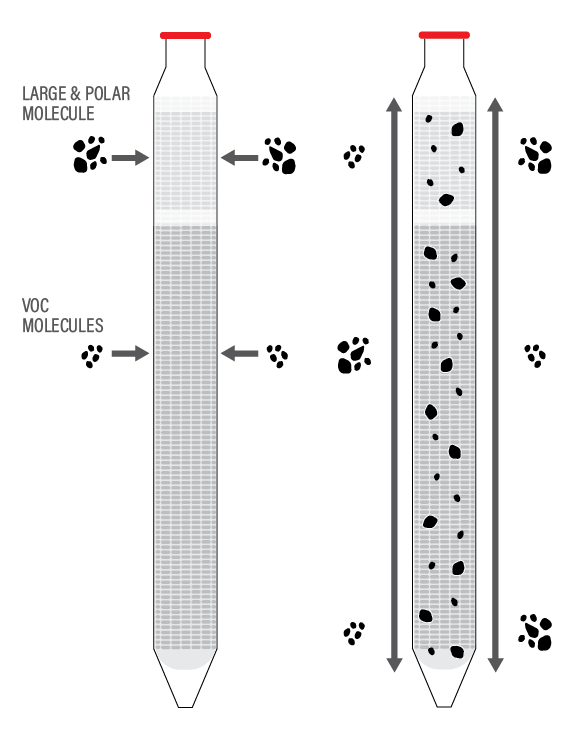
Large and polar molecules diffuse through the upper membrane and smaller molecules diffusion through the lower membrane. Molecules within the DMPDB diffuse throughout the water inside the sampler until concentrations within the DMPDB are in equilibrium with the surrounding water. Based on a diagram provided courtesy of Brad Varhol, President, EON Products.
Bench-Scale Testing
In 2017, a bench-scale study evaluated the ability of the DMPDBs to collect representative PFAS samples in a controlled environment using a polyvinyl chloride test chamber filled with a known volume of PFAS-free water that was spiked with 8 PFAS—including long-, medium-, and short-chain substances—at target concentrations of 20-30-ng/L. The liquid was mixed and allowed to stabilize for 6 days. The first test consisted of 9 samplers that were allowed to equilibrate with the tank water for 21 days (3 DMDPBs) and 41 days (6 DMPDBs) prior to sample collection.
A second test was performed for tank concentrations between 1-ng/L and 10-ng/L and a residence time of 21 days. A pair of control samples were taken from the chamber port during each retrieval event. All samples were analyzed for the 8 PFAS using a modified Method 537.
Field Study Methods
Side-by-side samples were collected from 10 wells with known PFAS impacts at a DoD facility using DMPDB and low-flow purge techniques. DMPDBs were filled with PFAS-free primer water, installed in one mobilization, and allowed to equilibrate for 21 days. This duration was applied based on the shorter of the two durations used during bench-scale tests (21 days and 41 days), where comparable results were observed. There is no maximum known timeframe for retrieval. A sample of the water used to fill the DMPDBs was collected to confirm it was PFAS-free. A single DMPDB was placed in the center of the screen of each well, and 2 DMPDBs were deployed in tandem in 2 of the wells.
After 21 days, the DMPDBs were retrieved from each well and sampled. High-density polyethylene tubing and a submersible pump were then deployed to purge and collect another sample using low-flow procedures. Low flow, DMPDB, and field quality control samples were packed in ice and shipped to a DOD-accredited analytical laboratory for analysis in accordance with Quality Systems Manual Version 5.3 Table B-15 for 24 PFAS.
Data Analysis
Some apparent stratification was observed in one of the two wells with tandem DMPDBs. As a result, only the lower DMPDBs were used for comparison to low flow samples for consistency. PFAS were reported in 126 pairs of the 240 pairs of results that were analyzed. All pairs with detections were used for comparison on a 1:1 regression plot, with low-flow results on the x-axis and DMPDB results on the y-axis. However, only pairs with results greater than five times the reporting detection limits were used for relative percent difference analysis to eliminate artificially high RPDs produced from comparing low values.
This process was also used to analyze results from a set of low-flow field duplicate samples collected during the field event for comparison, and for field study results less than 200 ng/L, which are more representative of values within the range of existing screening levels.
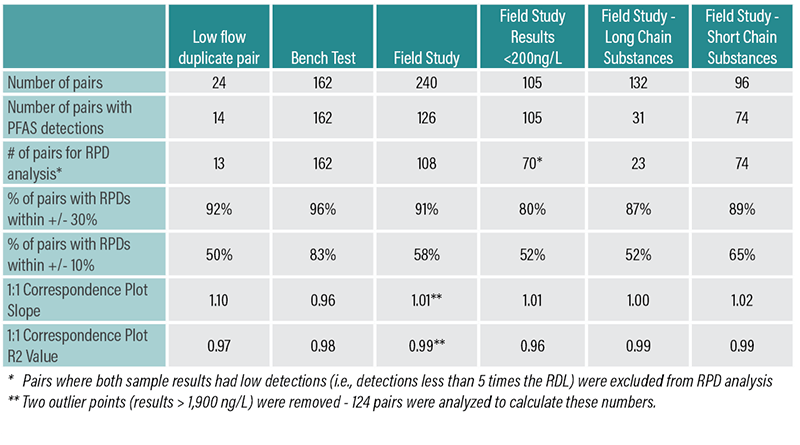
Statistical Results for the Bench Scale, Field Study, and Comparison Tests.
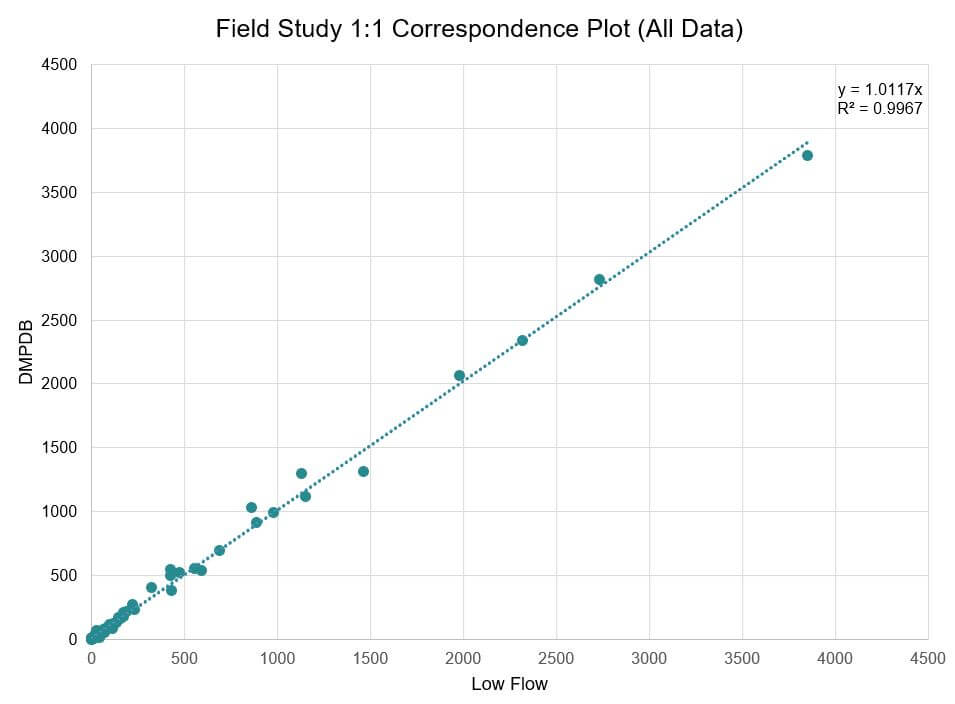
Paired PFAS detections from low flow and DMPDB samples analyzed in the field study
Real World Application
DMPDB PFAS results correlated well with the bench scale and field study samples. They were comparable to the low flow field duplicate sample and did not produce any results that affected comparisons to screening levels. Additionally, DMPDBs appeared to produce consistent results for both long- and short-chain substances, indicating that it may be suitable for PFAS sampling in future projects—although it may be better suited for sites with regular sampling events to eliminate the need for two mobilizations.
The cost benefits of DMPDBs were not evaluated in this effort. However, studies performed by the Interstate Technology & Regulatory Council in 2004 and Strategic Environmental Research and Development Program/Environmental Security Technology Certification Program in 2014 indicated that the cost savings associated with switching from low flow to equilibrium passive samplers can be in the range of 40-70 percent for long-term monitoring programs.
Future Research Efforts
Vertical stratification was observed in one of the wells but could not be fully understood without collecting additional tandem or duplicate samples. Given the nature of the sample collection process, where a sample is collected from the water column at equilibrium and at “natural conditions” without disturbance from pumping, there is a possibility of observing concentration variation within the well itself. This could be an area for future research.
An additional area of future research could be used to observe and quantify the impact of shortening equilibrium period of the DMPDB. During this study, a conservative period of 21 days was used based on the initial durations used at the bench scale test. Much shorter equilibrium periods are used for other analytes and could be evaluated with these samplers to provide more immediate results.
Disclaimer: Endorsement of this study or its results by the DoD is neither expressed nor implied.
By: Paul Caprio, P.G., Erica Thieleman, and Steven Gragert, CHMM (Military Munitions and Environmental Sciences Section, U.S. Army Corps of Engineers–Omaha District)
 For More Information Contact:
For More Information Contact:
Mahsa Modiri, PhD, PE
Director, Emerging Science and Innovation
Contact Us
